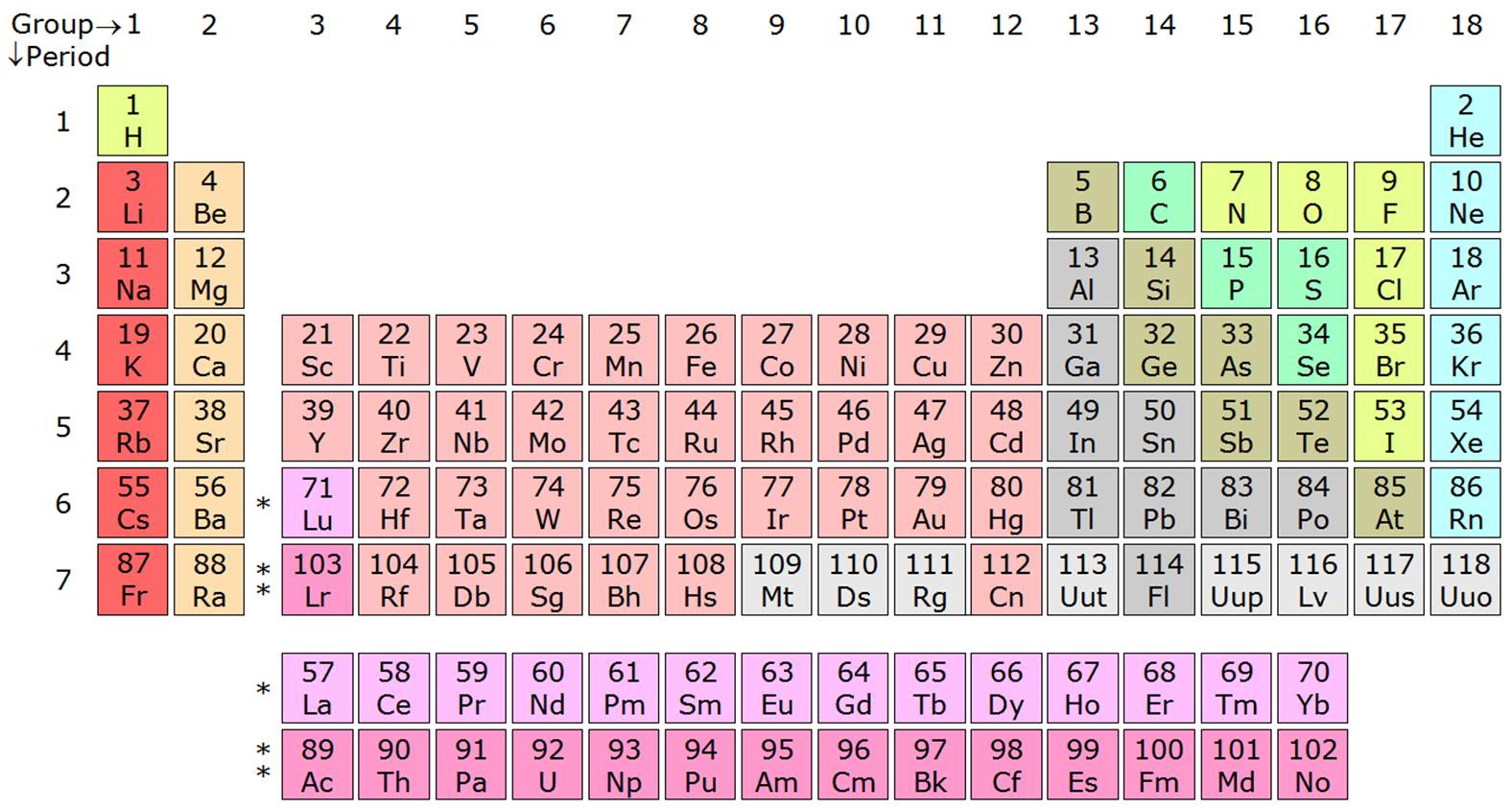
For example, in period 6 we have n going from 6, to 4, to 5 and back again to 4. And if one considers the long-form table based on an increasing value of n for each new row the situation is even worse. Admittedly there is a change in n from 2 to 3 as one crosses from Ne to Na in the left-step table, but this is better, not worse, than the change in n from 4 to 3 and back to 4 as one crosses, say, the fourth row in the conventional table. However, the principle of electron-shell filling follows an increasing rule for the quantum numbers n + l and not an increasing n rule. Olmstead suggests that the conventional, or medium-long form table, more clearly reflects the order of electron shell filling than the left-step form table. That is certainly a real break in properties.ĭr. Scerri's proposed table.Īs to the desirability of getting rid of the "implied" breaks between periods in the conventional table, both atomic radius and first ionization energy change immensely between any noble gas and its immediate successor alkali metal. Moreover, periodic trends such as first ionization energy and atomic size undergo striking changes between elements of group 18 (noble gases) and group 1 (alkali metals), yet these groups are adjacent in Dr. Scerri's proposed table not only shares this disadvantage but also divides the p block awkwardly. The left-step table can also be divided into analogous blocks and its columns labeled analogously, but its rows do not match up with highest principal quantum number (it changes from 2 to 3 between Ne and Na, and so on). In addition, the table itself becomes an important mnemonic tool for students. Inasmuch as the valence electrons are the most important determinant of the chemical properties of an element, this feature is the underlying explanation for periodicity. 
Labeling in this way is more than just place-holding. Only helium, which is unique because its s 2 valence configuration also is a closed shell, is "out of place" in this labeling scheme. 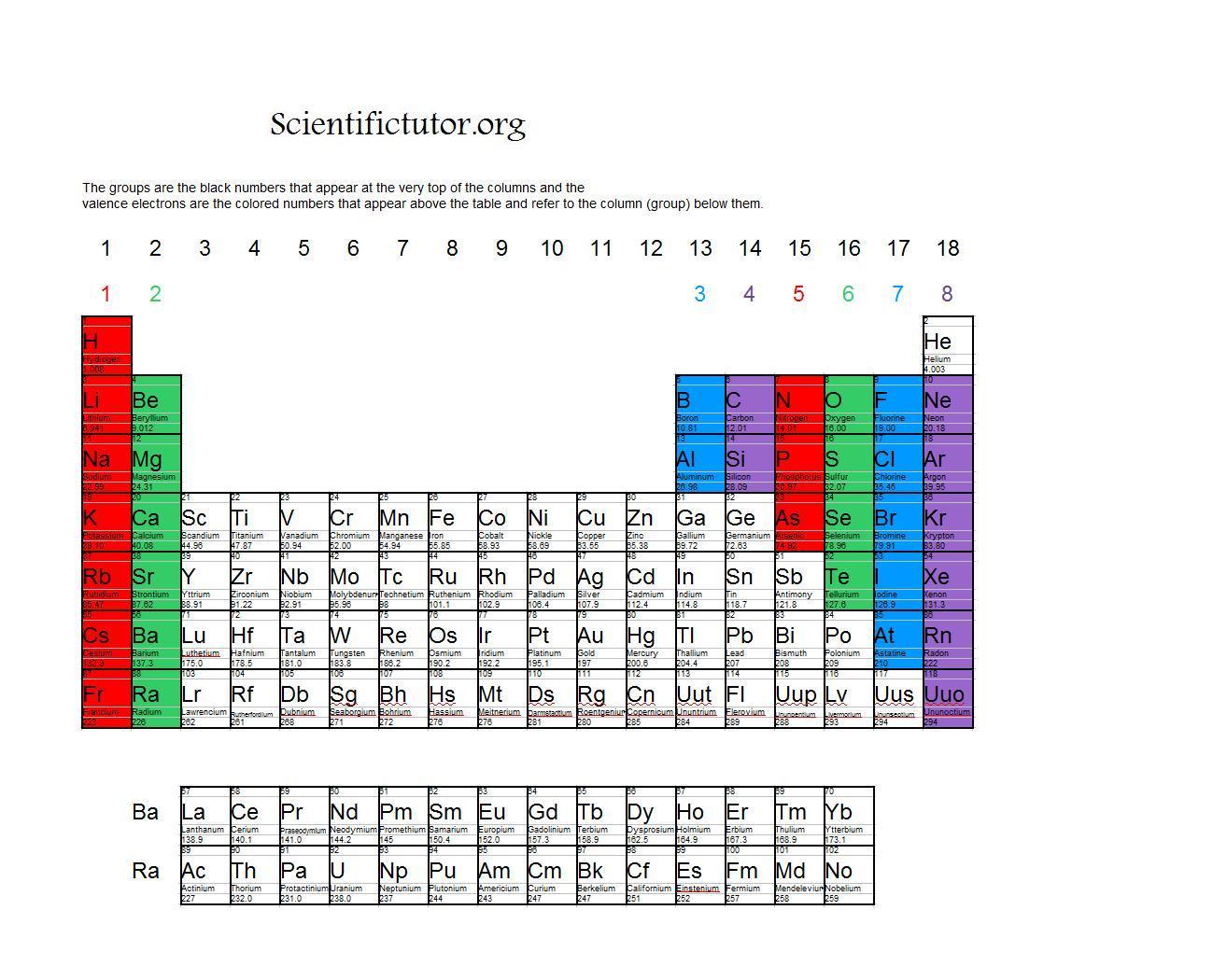
Further, each column of the table can be labeled with the number of valence electrons sharing that azimuthal quantum number, and each row of the table can be labeled with the highest principal quantum number found among the electrons. With reference to the valence electron configuration of the ground state of each element, the table can be viewed as consisting of rectangular blocks labeled by the azimuthal quantum number for the least stable electron (called s, p, d, f). 
Scerri's otherwise informative article about the periodic table ( "The Past and Future of the Periodic Table," January-February), an important underlying principle is not mentioned. Elements in each column have the same valence shell electron configurations, and the elements have some similar chemical properties.I am surprised that in Eric R. The same concept applies to the other columns of the periodic table. The organization of electrons in atoms explains not only the shape of the periodic table, but also the fact that elements in the same column of the periodic table have similar chemistry. Because much of the chemistry of an element is influenced by valence electrons, we would expect that these elements would have similar chemistry- and they do. They all have a similar electron configuration in their valence shells: a single s electron. Their electron configurations (abbreviated for the larger atoms) are as follows, with the valence shell electron configuration highlighted: Electrons, electron configurations, and the valence shell electron configuration highlighted. For example, take the elements in the first column of the periodic table: H, Li, Na, K, Rb, and Cs. If we look at just the valence shell’s electron configuration, we find that in each column, the valence shell’s electron configuration is the same. (The inner electrons are called core electrons.) The valence electrons largely control the chemistry of an atom. The electrons in the highest-numbered shell, plus any electrons in the last unfilled subshell, are called valence electrons the highest-numbered shell is called the valence shell. The periodic table is separated into blocks depending on which subshell is being filled for the atoms that belong in that section.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
